|
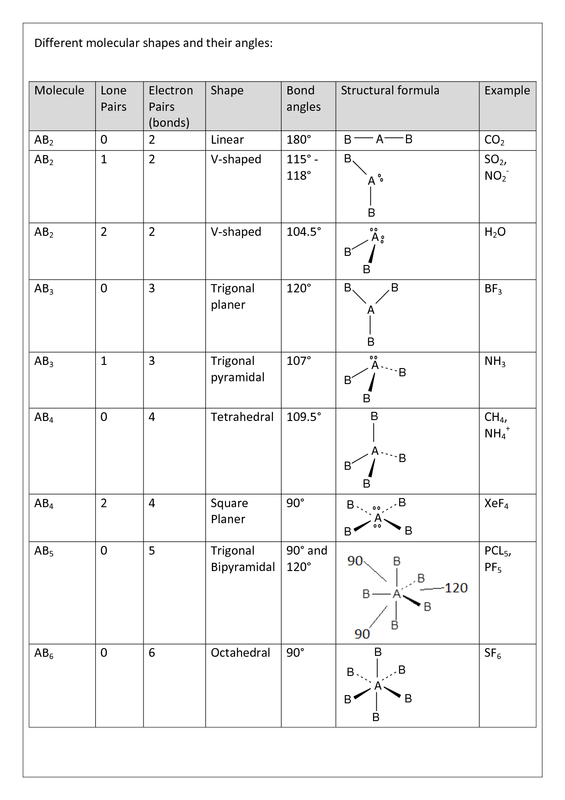
Be sure to account for any lone pairs surrounding the central atom to get a more accurate prediction. As they are 3D representations, dotted lines indicate bonds coming forwards out of the plane and wedges indicate bonds going away from the viewer. Lone pairs of electrons are indicated by bonds from the central atom with no terminal atom at the other end. We can also identify geometries by the steric number, which is the number of atoms bonded to the central atom. You can then compare this number with the chart to determine the molecular geometry and get an idea of the bond angles.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
